Nature actively bends the rules of time, offering distinct biological blueprints for defying death and decay. While you might expect every animal to slowly expire on a predictable biological clock, certain species bypass standard aging altogether through cellular resets, extreme metabolic pauses, or endless DNA repair mechanisms. By studying these extraordinary creatures, scientists unlock practical insights into longevity, cancer resistance, and regenerative medicine that could eventually transform human health. From sharks prowling the arctic depths for centuries to tiny jellyfish that reverse their own life cycles, you will discover nine animals that completely rewrite the rules of senescence. Dive into these extreme survival strategies and see exactly how wildlife outsmarts the aging process.

The Immortal Jellyfish Hits the Cellular Reset Button
In the warming waters of the Mediterranean Sea and oceans worldwide, a tiny, translucent creature silently defies the most fundamental rule of biology. The immortal jellyfish (Turritopsis dohrnii) measures barely 4.5 millimeters across, yet it wields an unparalleled survival mechanism. When confronted by starvation, physical injury, or sudden temperature drops, this hydrozoan does not simply die. Instead, it hits a biological reset button. Through a rare physiological process known as transdifferentiation, the adult medusa transforms its specialized cells back into unspecialized stem cells. It actively absorbs its own tentacles, sinks to the ocean floor, and reverts to a juvenile polyp colony. These polyps eventually bud off into dozens of genetically identical clones.
You can view this process as a butterfly reverting into a caterpillar just as it faces a predator. The National Geographic Society highlights how these tiny invertebrates continue to swarm the world’s oceans, often riding safely in the ballast water of heavy cargo ships. By observing transdifferentiation in the laboratory, medical researchers gain crucial insights into cellular reprogramming. Unlocking the precise genetic switches that allow the immortal jellyfish to rejuvenate its own tissue could one day lead to massive breakthroughs in human organ regeneration and targeted treatments for neurodegenerative diseases.

Greenland Sharks Outlast Empires in the Deep Cold
Far beneath the sea ice of the North Atlantic, the Greenland shark (Somniosus microcephalus) navigates the freezing darkness at an incredibly sluggish pace. Growing over 16 feet long, these apex predators hold the undisputed title of the longest-living vertebrates on the planet. By radiocarbon dating the crystalline proteins formed inside the sharks’ eyes before birth, researchers at the National Oceanic and Atmospheric Administration determined these creatures live at least 272 years. The oldest observed female reached an estimated age of nearly 400 years, meaning she swam the oceans before the Pilgrims landed at Plymouth Rock.
A massive body combined with a near-freezing habitat forces the Greenland shark’s metabolism to operate at a glacial pace. They grow less than a centimeter per year and do not reach sexual maturity until they hit 150 years of age. Often blinded by a highly specific marine parasite, they rely on a tremendous sense of smell to scavenge narwhals and beluga whales. Their exceptionally slow metabolic rate minimizes the production of damaging free radicals, preserving their cellular health over centuries. By analyzing the Greenland shark’s genome, scientists are discovering distinct DNA repair genes that actively suppress tumor growth and resist cardiovascular decay. Protecting these ancient sharks from commercial fishing bycatch remains critical, as their delayed maturity makes rebuilding depleted populations a multi-century endeavor.

Naked Mole-Rats Actively Suppress Cellular Aging
If you judge an animal purely by its physical appearance, the naked mole-rat (Heterocephalus glaber) might seem highly vulnerable. Hairless, practically blind, and equipped with massive protruding incisors, these subterranean rodents inhabit the arid deserts of East Africa. However, they possess a biological toolkit that constantly baffles geneticists. While mice and rats of similar size succumb to old age within three years, naked mole-rats frequently live past the age of 30. They exhibit negligible senescence; their bone density, cardiovascular health, and reproductive capabilities remain exceptionally robust throughout their entire lives.
The Smithsonian’s National Zoo reports that these creatures organize their deep burrowing colonies much like bees, with a single queen bearing young while sterile workers forage for roots and tubers. Their extreme longevity stems from a heavy, viscous sugar molecule in their tissue called high-molecular-weight hyaluronan. This substance actively prevents cells from crowding together, effectively stopping the formation of cancerous tumors before they even start. Furthermore, naked mole-rats thrive in low-oxygen tunnels by switching their metabolism to burn fructose, avoiding the severe cellular damage typically associated with oxygen deprivation. You can see why biomedical engineers study these rodents heavily; replicating their natural resistance to cellular degradation could drastically reshape human cancer prevention strategies.
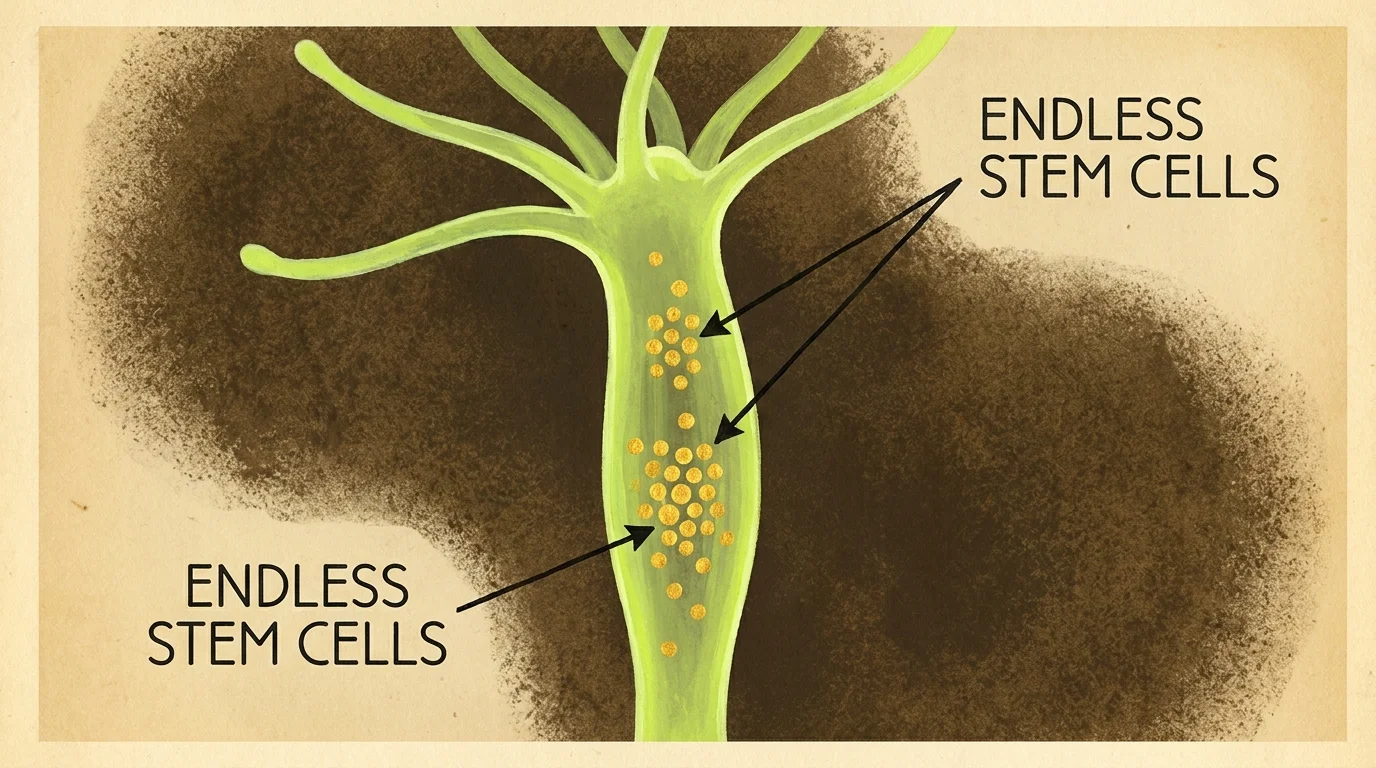
Hydra Maintain an Endless Supply of Stem Cells
Peer into the shallow edges of freshwater ponds and you might spot a Hydra (Hydra vulgaris). Resembling microscopic palm trees with tubular bodies and a crown of tentacles, these predatory invertebrates capture passing water fleas with specialized stinging cells. Biologists observing Hydra in isolated laboratory conditions discovered something astounding: their mortality rate does not increase with age. Whether a Hydra is two weeks old or ten years old, its probability of dying from natural biological decay remains practically zero.
This non-senescent lifestyle relies heavily on an aggressive, continuous cycle of cellular renewal. Stem cells compose the vast majority of a Hydra’s body. These unspecialized cells divide constantly, pushing older cells out to the extremities where they naturally slough off into the water. Within a span of just 20 days, a Hydra completely replaces every single cell in its physical structure. You can slice a Hydra into multiple pieces, and each fragment will rapidly regenerate into a completely new, healthy organism. Medical researchers routinely sequence the Hydra’s genome to isolate the specific transcription factors responsible for this endless stem cell production. Understanding how these tiny predators successfully avoid stem cell exhaustion delivers direct insights into halting human tissue degeneration and muscular atrophy.

Ocean Quahogs Record Centuries of Climate Data
Buried beneath the icy mud of the North Atlantic seabed, the ocean quahog (Arctica islandica) filters microscopic algae and quietly accumulates centuries of biological history. These edible bivalves look remarkably ordinary, yet their lifespans push the absolute limits of multicellular endurance. In 2006, scientists dredged an ocean quahog off the coast of Iceland. By counting the annual growth rings embedded within its hinge ligament, researchers concluded the clam—nicknamed Ming—lived for a staggering 507 years. Ming was born during the height of the Ming Dynasty in China and survived right through the global Industrial Revolution.
Ocean quahogs achieve this monumental lifespan through extreme metabolic stability and exceptional cellular defenses. Their cell membranes contain highly modified lipids that fiercely resist oxidation, preventing the steady accumulation of microscopic damage that normally ages an animal. Furthermore, researchers extract invaluable data from these long-lived clams. Because their shells add a distinct layer of calcium carbonate every single year, they trap precise isotopic signatures of the surrounding water. By studying quahog shells alongside modern carbon-dating techniques, you can read a continuous, year-by-year physical record of historical ocean temperatures and marine climate shifts.

Olms Survive Decades in Complete Darkness
Descend into the submerged limestone caves of the Dinaric Alps in southeastern Europe, and you enter the domain of the olm (Proteus anguinus). Locals historically referred to these pale, blind salamanders as “baby dragons” whenever heavy rains flushed them to the surface. Perfectly adapted to total darkness, olms navigate frigid underground streams using an acute sense of smell, highly sensitive underwater hearing, and specialized sensory organs that detect the electrical fields of their prey. Despite weighing less than an ounce, olms routinely live up to 100 years.
Their longevity directly correlates with a life spent in the absolute slow lane. The deep cave environment offers zero sunlight and severely limited food sources. In response, the olm drastically slows its metabolism and retains its juvenile gills throughout its entire adult life. When prey like aquatic snails or small crabs completely vanish, an olm can survive up to a decade without consuming a single meal; it simply reduces its activity to a standstill and safely reabsorbs its own tissue. Surprisingly, olms do not rely on excessive antioxidant production to stay young. Their mitochondria are highly efficient, generating energy without leaking the destructive free radicals that cause cellular aging. By studying the olm’s metabolic efficiency, researchers map new strategies for protecting human tissue during periods of starvation or severe physical trauma.

American Lobsters Produce an Anti-Aging Enzyme
The American lobster (Homarus americanus) crawls along the rocky ocean floors of the North Atlantic, snapping up crabs, mollusks, and sea urchins. Unlike humans, who stop growing after puberty and steadily lose muscle mass in their twilight years, lobsters exhibit indeterminate growth. They never stop growing, and their physical strength actually increases as they age. They also maintain their reproductive fertility until the very end of their lives, with older females producing vastly more eggs than younger counterparts.
This biological phenomenon hinges on a cellular enzyme called telomerase. Inside the nucleus of every cell, DNA is capped by protective structures called telomeres. Every time a human cell divides, its telomeres shorten until the cell eventually dies. Lobsters bypass this ticking biological clock entirely. Their tissues constantly produce telomerase, which meticulously rebuilds the telomeres and allows their cells to divide flawlessly over and over again. To determine their true age, scientists measure the growth bands hidden inside the lobster’s gastric mill, since the outer shell sheds constantly. Despite their cellular youth, lobsters do not live forever. As they grow immensely large, the process of shedding their massive exoskeletons requires an immense amount of metabolic energy. Eventually, older lobsters succumb to exhaustion during a molt or fall victim to shell disease. Observing lobster biology encourages oncologists to investigate how safely manipulating telomerase might prevent human cells from aging.

Tuatara Rely on Prehistoric DNA for Centenarian Lifespans
At first glance, the tuatara (Sphenodon punctatus) looks like an average lizard basking on a coastal rock. However, this olive-green reptile is the sole surviving member of the Rhynchocephalia order, a distinct evolutionary lineage that walked the Earth alongside the very first dinosaurs over 200 million years ago. Now restricted entirely to the predator-free offshore islands of New Zealand, tuatara routinely exceed 100 years of age.
Tuatara conduct their lives at a remarkably measured pace. They feature a highly unique dental structure—two rows of teeth in the upper jaw overlapping a single row on the bottom jaw—which they use to crush heavily armored beetles and weta insects. They actively hunt in temperatures as low as 40 degrees Fahrenheit, a chill that completely paralyzes most modern reptiles. They do not reach sexual maturity until they are 20 years old, and their bones continue to grow well into their late thirties. You can identify their most fascinating anatomical feature right on top of their heads: a parietal “third eye” complete with a lens and retina. While it does not process visual images, this eye detects ultraviolet light and helps the tuatara regulate its circadian rhythms and seasonal metabolic shifts. The International Union for Conservation of Nature closely monitors their survival, as studying these ancient survivors grants biologists an exclusive window into the anti-aging mechanisms of prehistoric wildlife.

Rougheye Rockfish Deploy Advanced DNA Repair
Hovering quietly above the deep-sea canyons of the North Pacific Rim, the rougheye rockfish (Sebastes aleutianus) navigates depths exceeding 1,500 feet, where the water temperature hovers near freezing. Dressed in vibrant reddish-pink scales, this species ranks among the longest-lived fishes ever recorded. Marine biologists verify their ages by counting the microscopic growth rings embedded within their otoliths—tiny calcium carbonate ear bones used for sensory balance. Through this precise method, researchers confirm the rougheye rockfish can live for at least 205 years.
Recently, geneticists sequenced the genomes of several rockfish species to pinpoint the exact evolutionary drivers of their immense lifespans. They discovered that long-lived rockfish species possess unique mutations directly inside their DNA repair pathways. Over a lifespan spanning two centuries, an animal accumulates a vast amount of genetic damage from natural radiation and everyday metabolic stress. The rougheye rockfish combats this decay by aggressively repairing double-strand DNA breaks. Furthermore, their immune systems contain expanded gene families that successfully suppress chronic inflammation. By reviewing the genomic architecture of the rougheye rockfish, genetic engineers can target the specific inflammatory responses that drive arthritis, neurodegeneration, and general aging in human populations.

Comparing the Longevity of Nature’s Outliers
To clearly see how these extraordinary creatures stack up against one another, review their maximum documented lifespans and the primary biological mechanisms they use to defy time.
| Animal Species | Native Habitat | Maximum Documented Lifespan | Primary Anti-Aging Mechanism |
|---|---|---|---|
| Immortal Jellyfish | Temperate and Tropical Oceans | Biologically Immortal | Transdifferentiation (Cellular Reset) |
| Hydra | Freshwater Ponds and Streams | Biologically Immortal | Endless Stem Cell Renewal |
| Ocean Quahog | North Atlantic Seafloor | 507 Years | Oxidation-Resistant Cell Membranes |
| Greenland Shark | Arctic and North Atlantic Oceans | 400+ Years | Extreme Metabolic Suppression |
| Rougheye Rockfish | North Pacific Deep-Sea Canyons | 205 Years | Advanced DNA Repair and Anti-Inflammation |
| American Lobster | North Atlantic Coastline | 140+ Years | Continuous Telomerase Production |
| Tuatara | New Zealand Offshore Islands | 100+ Years | Delayed Maturity and Cold Tolerance |
| Olm | European Subterranean Caves | 100 Years | Mitochondrial Efficiency and Starvation Resistance |
| Naked Mole-Rat | East African Deserts | 30+ Years | High-Molecular-Weight Hyaluronan |
The Future of Biological Discovery
Investigating the animals that age differently from the rest of the natural world does significantly more than satisfy scientific curiosity. Their bodies hold the functional blueprints for defeating the most common cellular threats facing humanity today. When you look at a naked mole-rat actively resisting cancer, a rockfish repairing its own DNA, or a tiny jellyfish completely reversing its biological clock, you witness millions of years of evolutionary trial and error resulting in brilliant genetic efficiency. Protecting these unique species from habitat destruction, climate shifts, and commercial overfishing remains an absolute necessity. If these creatures disappear from our deep oceans and subterranean cave systems, their genetic secrets will vanish permanently with them. Preserving global biodiversity actively safeguards the medical breakthroughs of tomorrow, proving that nature remains our greatest and most experienced teacher in the ongoing quest for human longevity and lasting health.















